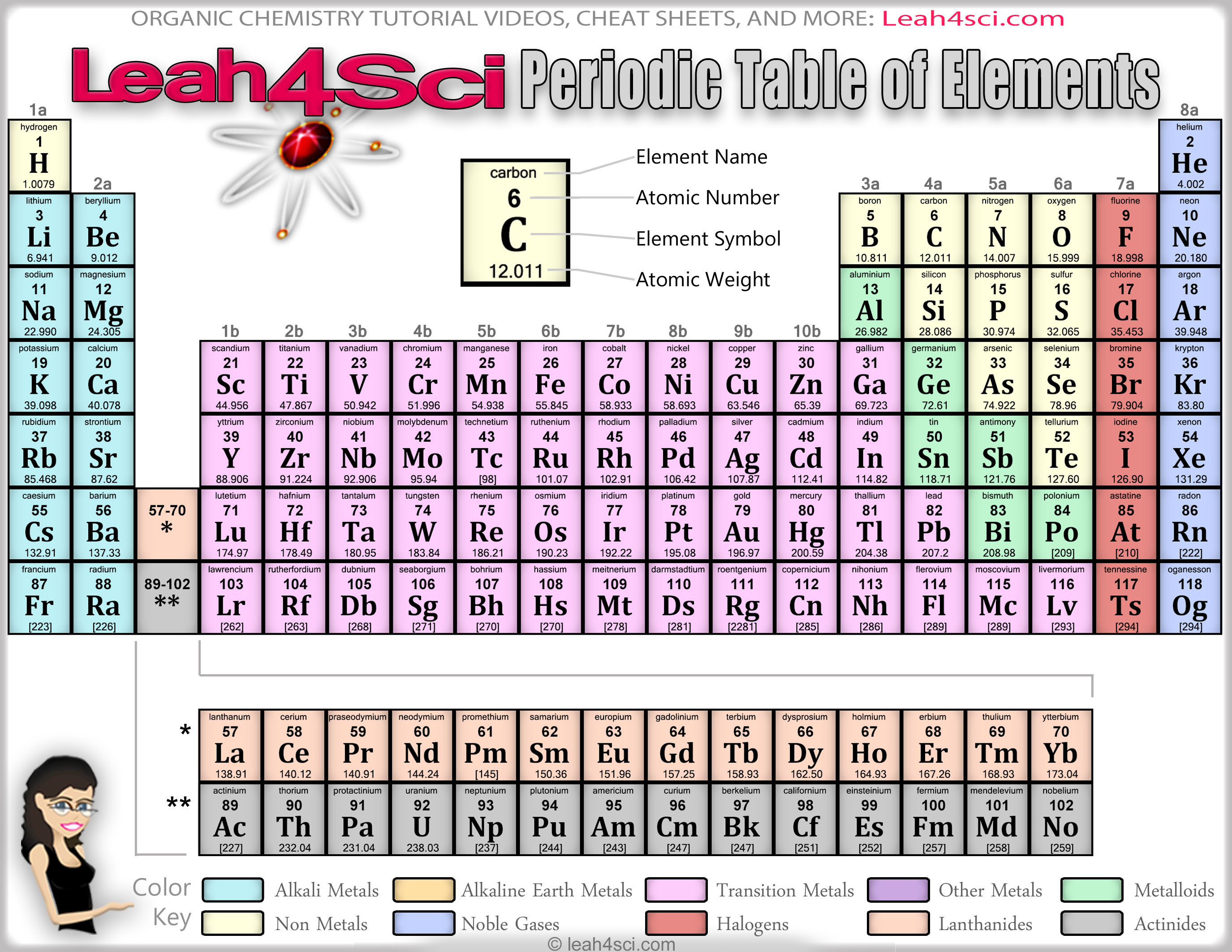 
Read about electron configurations to learn more. Here you’ll find a link to a downloadable periodic table with charges. This gives them a full S subshell, which is a pretty stable configuration. For example, metals often lose enough electrons to leave two valence electrons remaining (example: carbon family with +2 charge). Most elements have the ability to form more than one charge, but there are patterns to be found there, as well.You can check out this fancier Periodic Table to see the most common charges of most of the elements.Therefore, sometimes that row is thought of as having “+/- 4” charge, which fits nicely between the +3 and -3 columns. But it’s possible for carbon and silicon to form a C 4- / Si 4- ions, and all of that family’s elements can form +4 ions. The elements in the carbon family aren’t listed as +4 or -4 charged ions, because they are more likely to form covalent bonds instead of being ions.For example, iron can form Fe 2+ and Fe 3+ ions, depending on the situation. The transition metal elements (middle section of the table) don’t have the same type of predictable charge patterns, and most of them are capable of forming more than one type of ion.But at least we can say that it’s capable of forming a P 3- ion to fit the pattern shown in the chart. For example, phosphorus is actually more likely to be found with a positive charge in a compound with oxygen. Atomic size depends on the nuclear charge as well as the number of shells. The Group 8A elements already have eight electrons in their valence shells, and have little tendency to either gain or lose electrons, and do not readily form ionic or molecular compounds. Effect of Nuclear Charge on Atomic Size in the Periodic Table. These elements don’t always follow such this pattern. That is, the Group 7A nonmetals form 1- charges, the Group 6A nonmetals form 2- charges, and the Group 5A metals form 3- charges.In the periodic table, the vertical columns are called groups and the horizontal rows are called periods. (The numbers at the top of the columns show what charge that family’s elements sometimes have as ions.) Periodic Table of Elements - The periodic table is the tabular arrangement of all the chemical elements on the basis of their respective atomic numbers. Halogens have 7 valence electrons, and they gain one more electron, filling their valence shells to form ions with -1 charge.A trend is to find the charge by looking at the Group on the Periodic. For example, iron(II) has a 2+ charge iron(III) a 3+ charge. Single-Replacement- A free uncombined element replaces another combined element in a. When writing formulas for ions and ionic compounds we often need to find the ionic charge. Roman numeral notation indicates charge of ion when element commonly forms more than one ion. Alkaline earths have two valence electrons, which they lose to form ions with +2 charge. Ionic Charges of the Periodic Table: Valence electrons are the Grade 10.Alkali metals all have one valence electron, which they lose to form ions with +1 charge.(Disclosure: there are many exceptions to this behavior)Įxamples (see Element Families notes if needed): Metals tend to lose all of their valence electrons, and nonmetals tend to gain enough electrons to get to 8 valence electrons for a full shell. The feature is a testament to the scientific research done at the lab, but also. Idaho National Laboratory has enlisted its experts, researchers and writers to produce an online interactive Periodic Table that offers pop-up information on every single known element. /PeriodicTableCharge-BW-56a12db13df78cf772682c34.png)
the ions formed are negative, because they have more electrons than protonsįor elements in groups 6 and 7, the charge on the ion is equal to (8 minus group number).You can use a Periodic Table to find the charge that an element is likely to have as an ion.Įlements’ position on the table tells you their valence electrons, and that determines what charge would give them full valence shells as as ions. INL INTERACTIVE PERIODIC TABLE OFFERS DETAILED INFORMATION ON ALL 118 ELEMENTS.The outer shells of non-metal atoms gain electrons when they form ions: A sodium atom loses one electron to form a sodium ion Forming negative ions the ions have the electronic structure of a noble gas (group 0 element), with a full outer shellįor elements in groups 1, 2 and 3, the number of electrons lost is the same as the group number.the ions are positive, because they have more protons than electrons.Metal atoms lose electrons from their outer shell when they form ions: non-metal atoms gain electrons to form negatively charged ions.metal atoms lose electrons to form positively charged ions.Ions form when atoms lose or gain electrons to obtain a full outer shell: If you need to go back and review any of the topics again, use the timeline. elementary charge electron energy proton mass fine-structure constant 1/137.035 999 Rydberg constant 10 973 731.569 m1 Boltzmann constant 1. An ion is an atom or group of atoms with a positive or negative charge. Watch videos about Dmitri Mendeleev, electronegativity and effective nuclear charge.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
